Biomedical Research
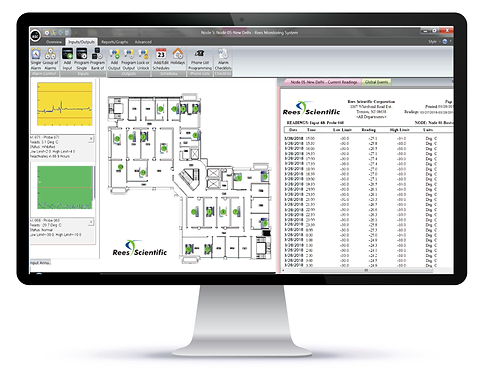
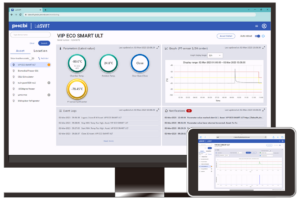
Rees Scientific’s Monitoring System protects your critical research, equipment and precious commodities. Since 1982, Rees Scientific has been the industry standard for automated temperature monitoring. We monitor equipment and parameters such as temperature, humidity, light, differential pressure, air changes per hour and more. Rees Scientific offers a redundant monitoring system designed to meet your critical AAALAC, CLIA and GxP regulations. The Rees System provides automated daily printouts, alarm notification, audit trails, and complete data encryption. You get alarm notification via local sonic, interactive dialed telephone, escalating email and text messaging, at any time of the day or night.
Pharmaceutical and Biotechnology
Building on over four decades of experience in the Pharmaceutical industry, Rees Scientific is the preferred monitoring solution for pharmaceutical research, manufacturing, storage and lab animal research. Rees Scientific’s data logging modules are a simple and easy way to monitor your critical equipment and parameters. Rees Scientific can help you prepare for GMP and GLP compliance standards. As an ISO 9001 certified & 17025 accredited Company, we are constantly upgrading policies & procedures to comply with today’s highly regulated environments. Rees Scientific’s service and engineering team can partner with you to implement the best system according to your requirements. Regardless of your location, their service and 24/7/365 technical support teams can ensure needs are responded to promptly.
Blood and Tissue Banking
With 40 plus years of experience, Rees Scientific has positioned itself as the leader in Blood Bank monitoring worldwide through continuous product development and technical innovation. You can reduce cost and guarantee AABB, FDA, CAP and Joint Commission compliance with Rees Scientific. Rees Scientific continuous monitoring systems were developed specifically with hospital blood banks in mind. In meeting today’s toughest standards for monitoring and total support documentation, Rees Scientific systems can turn any hospital blood bank into a complete command and information center. In addition to keeping your products safe 24/7/365 with the data logging modules, Rees Scientific continuous monitoring systems provide up to date information on your entire lab. Whether a routine report or specific unit interrogation, every Rees Scientific System report gives you detailed, easy to understand and accurate accounts of all transactions within your monitoring system.
Hospitals
From Pharmacy to Blood & Tissue Banks to Core Labs & Nutrition, Rees Scientific can help you monitor multiple departments. We can help protect critical assets in cell therapy, stem cells and regenerative medicine. Since 1982, Rees Scientific has been the most compliant continuous environmental monitoring system in the industry. We monitor equipment such as refrigerators, freezers, stability chambers, cryogenics, and cold storage. We monitor parameters such as temperature (+1300 to -196 °C), humidity, light, differential pressure and much more. Automating environmental monitoring increases accuracy, provides early warnings to address problems before they occur. Meet compliance for Joint Commission, USP <797>, USP <800>, DOH and much more.
Pharmacy
Rees Scientific provides continuous automated monitoring for the Pharmacy industry with a FDA compliant monitoring system. It helps monitor critical assets, equipment, and environments such as cleanrooms and compounding pharmacies. The Rees system can monitor temperature of any cold storage (refrigerators, freezers, ultra-low freezers) from +1300 to -196 °C. Rees Scientific’s Monitoring System can help monitor the temperature of crucial vaccines and medications. It can monitor a diverse facility with multiple locations while providing immediate data access and sophisticated analysis from any desktop computer, tablet, or phone. Instant alarm notifications are available via local sonic, interactive dialed telephone, escalating email and text messaging, at any time of the day or night. The Rees System provides automated daily printouts, alarm notification, audit trails, and complete data encryption. These standard features help organizations meet compliance for USP <797>, USP <800>, FDA, ASHP, GMP, Joint Commission, and other regulatory requirements.
Repositories
Repositories often handle millions of specimens including cell banks, tissue, sera, plasma, slides and nucleic acids. Freezer alarms, on- and off-site notification, documentation and validation are critical components of a successful repository. As with most businesses, repositories are seeking a competitive edge when bidding on large contracts. Having Rees Scientific as a partner can help your organization complete its mission statement and provide quality storage for your clients’ valuable and often irreplaceable specimens.
Forensic Labs
Requirements for storage, monitoring, and data collection of DNA evidence has increased in the last decade. The media attention and public awareness of the role of crime labs has increased dramatically. Though ASCLD/LAB accreditation is voluntary, many defense attorneys will challenge evidence if a lab is not accredited. ASCLD/LAB accreditation demonstrates that a crime laboratory’s procedures meet established standards. A quality assurance program is a vital part of achieving and maintaining accreditation. Installation of the Rees monitoring system ensures that no evidence has been lost or degraded and is a valuable tool to achieve quality assurance and operations in evidence processing and storage.
In Vitro Fertilization
In Vitro Fertilization is a very personal and complicated science that requires accurate and reliable equipment monitoring and alarming. As couples navigate the delicate journey of infertility, IVF facilities must ensure that their client’s precious cargo (embryos, sperm, eggs) are safe and protected. Both incubation and cryogenic preservation are the lifelines of this industry. The slightest variations in environmental factors such as temperature, humidity, LN2, O2, and CO2 concentration can literally mean the difference between life and death. Immediate notification allows your staff to address these changes and take action before they put future lives in jeopardy. Rees Scientific can provide you with the necessary protection needed to allow your organization to focus on your patients.